Bromine- Periodic Table of Videos
All About Bromine
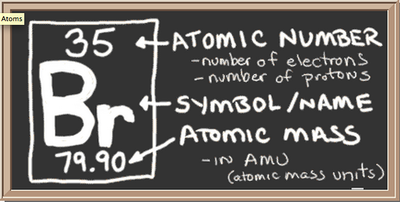
Bromine is number 35 on the periodic table. Its atomic number is 35 because it has 35 protons in its nucleus. Bromine also has 35 electrons. It has 35 electrons because electrons balance out protons in order to give the element a neutral charge. Its atomic symbol is Br. The atomic mass of Bromine is 79.904. Bromine is in series 17, which means it has 7 valence electrons. Bromine is in group 17 because it is a halogen. It is located in series 17 along with the other 4 halogens. Bromine also has 4 energy levels. There are about 45 neutrons in Bromine. Bromine is a nonmetal.
Bromine Electron Dot Diagram

Since Bromine is in group, or series, 17, it has 7 valence electrons. Group 1 on the periodic table has 1 valence electron. Group 2 on the periodic table has 2 valence electrons. The amount of valence electrons in groups 3-12 cannot be predicted because their oxidation numbers are unknown. To find the number of valence electrons in groups 13-18, you take the series number and subtract 10 from it. Therefore, if Bromine is in series 17, 17-10 is 7, so Bromine has 7 valence electrons. The electron dot diagram to the left depicts the number of valence electrons bromine has. The dots are arranged in a specific pattern to show how many valence electrons Bromine has.
"35 Br - Bromine." AIS Physical Science. N.p., n.d. Web. 06 Jan. 2015.
"Electrons In The Shells." Chem 4 Kids. N.p., n.d. Web. 06 Jan. 2015.
"Plz Explain Lewis Dot Structure and Electron Dot Structure of Each Elementexplain All Things Draw Figure - Homework Help - ENotes.com." Enotes.com. Enotes.com, n.d. Web. 06 Jan. 2015.
"Electrons In The Shells." Chem 4 Kids. N.p., n.d. Web. 06 Jan. 2015.
"Plz Explain Lewis Dot Structure and Electron Dot Structure of Each Elementexplain All Things Draw Figure - Homework Help - ENotes.com." Enotes.com. Enotes.com, n.d. Web. 06 Jan. 2015.